By Bob Jones
Rock & Gem continues to celebrate the chemical elements since the United Nations declared 2019 the International Year of the Periodic Table of Chemical Elements. The discovery of many of the elements during the last five centuries is fascinating and as full of detective suspense as any murder mystery.
In the On The Rocks column of the March issue, we described how Hennig Brand, in the mid-1560s, accidentally discovered phosphorus while searching for the elixir of life. This was the first element extracted from a compound. By that time the alchemists studying matter were changing and the modern science of chemistry was emerging. Scientific principles were being developed and the search for elements became a formal not accidental scientific quest.
By the 1500s we already knew of a number of naturally occurring elements. Gold, copper, mercury and several others were found in their natural state though they were not recognized as true elements that could not be changed through ordinary chemical means.
Changes Due to Chemistry
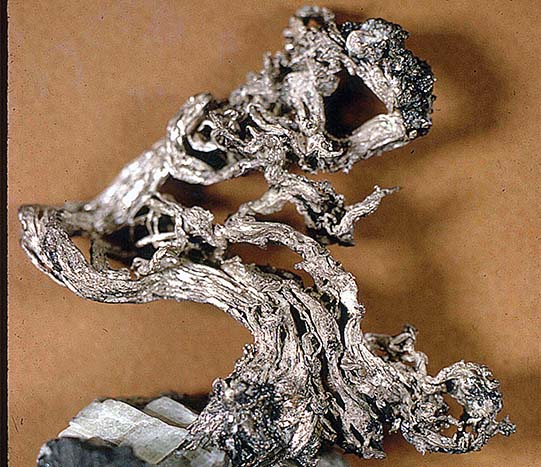
While gold, copper, and silver were found in their natural form on earth, two of the earth’s very common metals, aluminum (Al#13) and zinc (Zn#30) are not. Aluminum is the basis for the common mineral family, and the feldspars make it the most common metal in the earth’s crust. Yet it remained hidden in pure form until the mid-1800s. When finally produced, aluminum was so rare it was more valuable than gold. It was struck into coins like gold and silver and displayed with the French crown jewels. My how things have changed thanks to chemistry!

Zinc is the fourth most common metal in the earth’s crust preceded only by aluminum, iron, and copper. We get most of our zinc from the zinc sulfide sphalerite and several more beautiful secondary species like smithsonite, zinc carbonate, and hemimorphite, and zinc silicate hydroxide hydrate.
Today we have many uses for zinc. The uses include coating steel and iron to prevent oxidation or rust, and alloying with copper to form brass. We use zinc in the electronics industry, and every breath you take depends on zinc in a compound, because it is important in removing unwanted carbon dioxide from your lungs!
Zinc minerals have been around since the earliest days of mining. The great silver mines of Laurium (Lavrium), Greece supplied silver so the Greeks could build ships to defeat Xerxes over 2,000 years ago. Incredibly, smithsonite from this ancient deposit is still seen for sale today.
Impact of Zinc Extraction
However, zinc does not occur naturally on earth, only in compounds. But according to various historical documents, apparently, zinc had been extracted in its ‘pure’ form in 1347, in the country of India. It’s believed, the Chinese probably smelted it first, most likely from smithsonite, which is one of the easiest zinc minerals to smelt. The process of obtaining zinc through smelting was even documented by the Chinese. Suffice to say the Chinese were eons ahead of the Europeans in many ways at that time. They invented paper, the seismograph, the compass, gunpowder, and a few other useful things long before Europe came out of the Dark Ages!
It was not until the early 1700s that European scientists began smelting calamine (hemimorphite) to obtain zinc, which was initially a waste by-product of smelting. Andreas Marggraf is credited with the scientific extraction of zinc element. He combined calamine with charcoal in a sealed vessel, heated it intensely, and obtained pure zinc that could be scientifically tested to determine the true chemical properties. Only then was zinc deemed to be an element and could take its place on the periodic table.
Of all the elements discovered through scientific research and experiments, the oddest one has to be germanium (#32). You may recall from reading my column in the March issue that germanium was unknown at the time Dimitri Mendeleev developed his first Periodic Table of Chemical Elements in the mid-1800s. As he worked, Dimitri recognized that elements could be grouped by their similar chemical properties into certain families. As he listed the known elements by their properties, he left spaces for the elements he thought ought to be there based on properties of known elements. He even gave names to three missing elements, ekaboron, ekaaluminum and ekasilicon, which were discovered at a later date. Ekaalminum was eventually found and named gallium (Ca#31). Ekaboron was eventually discovered to be scandium (Sc#21). Ekasilicon turned out to be germanium (Ge#32). The first two were discovered as solids and were easily observed and collected. Germanium is normally a liquid at room temperatures, which Mendeleev predicted, and therein lies the mystery of its discovery.
It all began when scientists were examining the complex silver ores from the Himmelsfurst mine, near Freiberg, Saxony, which had been worked since its discovery a thousand years earlier. These ores are rich in silver and a variety of other elements. It was not until the 1880s that a new mineral appeared in the Himmelsfurst mine ores and was named argyrodite. At the time this new mineral’s chemistry was unknown and became the subject of a study by German Professor H.S. Richter. He was able to identify silver and sulfur in the new mineral, but more work was needed.
Furthering Silver Ore Exploration
Clemens A. Winkler took over the process to obtain a complete analysis of the new

mineral argyrodite. Winkler searched the new mineral for antimony, tin and other known elements. Once he had extracted every known element from the ore what remained had to be the missing component, the new element. He evaporated all the remaining solution, but when he checked his apparatus there wasn’t anything there! Whatever had been there had disappeared! How could that be? Undaunted he spent four more months working on the material, retorting, evaporating, chemically treating, filtering on and on without success.
Then one morning as he walked into his laboratory he happened to glance up at the ceiling. The ceiling was covered with a white powder that should not have been there. It was the missing material. It had turned to a liquid during his experiments just as Dimitri had predicted. It had evaporated unseen and condensed on the ceiling. The white powder contained the new element, which was easily extracted in pure form. Winkler had finally found germanium!
You might ask if Winkler’s endless search was worth it? Of what use was germanium? It seemed to have none and was considered just another element. But in World War II we found a use for it in making semiconductor diodes, and transistors, among other devices. Germanium suddenly became very useful and still is as a component of modern electronics.
Do you realize that if you lived in the early 1700s and suddenly died it could have been from lack of dephlogisticated air? A prevailing theory at the time was that objects could only burn and animals only breathed because of phlogiston in the air. Luckily, there was another school of thought that argued there was some unknown element in air, not phlogiston, which was needed. Phlogiston is a word from the Greek meaning “burning up.” The thinking was that once the phlogiston was depleted in the air, burning and breathing stopped.
Understanding the Oxygen Theory
The theory scientists had, and was proved to be right was the oxygen theory. Oxygen was necessary for burning to happen. The problem was that no one had isolated oxygen or phlogiston and neither theory could be proven. Scientists in several countries, including England and Sweden, worked diligently to prove the existence of the life-giving gas and debunk the phlogiston theory. We now know the gas is oxygen. Where would we be without oxygen to form many minerals?
Englishman Joseph Priestley is credited with producing the first sample of oxygen, although it’s reported he thought he had produced phlogiston. Just before Priestley discovered his life-giving gas, Swedish druggist Carl Scheele had done the same thing. Scheele had produced oxygen but did not publish his findings. Priestley published his findings and in turn received the credit. Scheele showed his gas could keep a candle burning. Priestley, on the other hand, not only showed his gas would keep a candle burning but would also keep a mouse alive. Oddly, Priestley chose to call his gas dephlogisticated air. It was not until the French scientist Antoine Lavoisier, the Father of Modern Chemistry actually identified the gas and named it oxygen.
Lavoisier was brilliant. He proved sulfur was an element and predicted the properties of the then unknown element that is so important in the electronics industry today, silicon (Si#14). Lavoisier was also sure that Priestley’s dephlogisticated air was necessary for the formation of all acids. He was wrong but decided to give Priestley’s gas a name based on Greek ‘oxys,’ which means sharp or acid, and mistakenly named the life-giving gas “acid former.”
Unfortunately, Lavoisier, a true genius, was a nobleman during the French Revolution. His government job was as a tax collector. When the Revolution got going all tax collectors were judged to be “stealing from the citizens” so all were condemned to the guillotine, including Lavoisier.

Highlighting Helium
There is one element, helium, which was first found in space, not on earth. It was recognized in the gaseous corona of the sun. Now we find it in quantity on earth as one of the many components in natural gas found in petroleum deposits. When someone hands you a floating balloon you are actually holding a container of helium. If you breathe it in your voice sounds funny because the density of helium is less than air. We use it in lighter-than-air ships and in other ways, like food preservation to keep oxygen away from the food, especially fruit.
Norman Lockyer first suggested helium as being in the sun’s atmosphere but many laughed him at. Later, he was vindicated when he studied a radioactive mineral and found helium as one of the mineral’s components. By the 1920s, we were finding it in quantity as one of the gases in natural gas. We now know it is the lightest of the Noble gas elements and is not flammable.
Helium was just one of the gases in the sun’s atmosphere. Scientists began to identify elements in the sun, of which there are dozens, by using a prism or other device to spread sunlight in the same manner as a rainbow. In studying the light spectrum in sunlight German scientist, Joseph von Fraunhofer among others recognized dark lines in the sun’s color spectrum. These were later realized to be absorption lines caused by gases of different elements in the sun’s atmosphere. These gases were absorbing certain light waves emitted by different elements in the sun’s atmosphere. We could duplicate the lines here on earth proving the sun was producing those same elements. This led to another avenue of research into the origin of elements in space and here on earth.
More interesting stories of the search for the chemical elements will be described in a future On the Rocks column.
Author: Bob Jones
 Holds the Carnegie Mineralogical Award, is a member of the Rock-hound Hall of Fame, and has been writing for Rock & Gem since its inception.
Holds the Carnegie Mineralogical Award, is a member of the Rock-hound Hall of Fame, and has been writing for Rock & Gem since its inception.
He lectures about minerals, and has written several books and video scripts.